Nuper, JAMA Oncology (IF 33.012) magnum investigationis exitum [1] divulgavit, a turma Professoris Cai Guo-ring ex Nosocomio Cancri Universitatis Fudan et Professoris Wang Jing ex Nosocomio Renji Universitatis Jiao Tong Scholae Medicinae Shanghaiensis, una cum KUNYUAN BIOLOGY: "Detectio Praecox Morbi Residui Molecularis et Stratificatio Periculi pro Cancro Colorectali Stadii I ad III per Methylationem DNA Tumoris Circulantis et Stratificationem Periculi". Hoc studium est primum studium multicentricum in mundo quod technologiam methylationis multigeni ctDNA sanguinis fundatam in PCR ad praedictionem recidivationis cancri colorectali et monitorationem recidivationis adhibet, viam technicam et solutionem magis sumptu-efficacem praebens comparatam cum methodis technologiae detectionis MRD existentibus, quae exspectatur usum clinicum praedictionis et monitorationis recidivationis cancri colorectali magnopere emendare, et superviventiam aegrotorum et qualitatem vitae significanter emendare. Studium etiam a periodico eiusque editoribus magni aestimatum est, et in hoc numero inter commendationes praecipuas numeratum est, et Professor Ioannes Ruiz-Bañobre ex Hispania et Professor Ajay Goel ex Civitatibus Foederatis Americae ad id recensendum invitati sunt. Studium etiam a GenomeWeb, praecipuo medio biomedico in Civitatibus Foederatis Americae, nuntiatum est.

Cancer colorectalis (CCR) est tumor malignus tractus gastrointestinalis communis in Sinis. Data ab Instituto Internationali Investigationis Cancri (IARC) anno 2020 ostendunt 555,000 novos casus in Sinis fere tertiam partem mundi repraesentare, cum incidentia ad secundum locum inter cancros communes in Sinis ascendens; 286,000 mortes fere tertiam partem mundi repraesentant, quinta causa frequentissima mortuum ex cancro in Sinis obtinens. Quinta causa mortis in Sinis. Notandum est inter aegros diagnositos, stadia TNM I, II, III et IV 18.6%, 42.5%, 30.7% et 8.2% respective esse. Plus quam 80% aegrotorum in stadiis mediis et posterioribus sunt, et 44% eorum metastases distantes simultaneas vel heterochronicas ad iecur et pulmonem habent, quae graviter tempus supervivendi afficiunt, salutem incolarum nostrorum periclitantur et grave onus sociale et oeconomicum afferunt. Secundum statistica Centri Nationalis Cancri, incrementum medium annuum sumptus curationis cancri colorectali in Sinis est circiter 6.9% ad 9.2%, et sumptus personales valetudinis aegrotorum intra unum annum a diagnosi usque ad 60% reditus familiaris occupare possunt. Aegroti cancro affecti morbo laborant et etiam magna pressione oeconomica subeunt [2].
Nonaginta centesimae laesionum cancri colorectali chirurgice removeri possunt, et quo citius tumor detegitur, eo altior est quinquennalis superviventia post resectionem chirurgicam radicalem, sed generalis recidivationis proportio post resectionem radicalem adhuc est circiter 30%. Rationes quinquennale superviventiae cancri colorectali in populatione Sinensi sunt 90.1%, 72.6%, 53.8% et 10.4% pro stadiis I, II, III et IV, respective.
Morbus residuus minimus (MRD) causa maior est recidivationis tumoris post curationem radicalem. Recentibus annis, technologia detectionis MRD pro tumoribus solidis celeriter progressa est, et plura studia observationalia et interventionalia gravia confirmaverunt statum MRD postoperativum periculum recidivationis postoperativae cancri colorectalis indicare posse. Probatio ctDNA commoda habet quod est non invasiva, simplex, rapida, cum alta accessibilitate exemplorum et superans heterogeneitatem tumoris.
Litterae Civitatum Foederatarum Americae NCCN de cancro coli et litteris Sinensibus CSCO de cancro colorectali ambae statuunt probationem ctDNA, ad determinationem periculi recidivationis postoperativae et selectionem chemotherapiae adjuvantis in cancro coli, informationem prognosticam et praedictivam praebere posse ad decisiones de curatione adjuvante pro aegrotis cum cancro coli stadio II vel III adiuvandas. Attamen, pleraque studia exstantia in mutationibus ctDNA intendunt, quae innituntur technologiae sequentiationis altae capacitatis (NGS), quae processum complexum, longum tempus praeparationis, et sumptum altum habet [3], cum levi defectu generalizabilitatis et praevalentia humili inter aegrotos cancro.
In aegrotis cancri colorectali stadii III, monitorium dynamicum ctDNA per NGS usque ad $10,000 pro singula visitatione constat et tempus exspectationis usque ad duas hebdomades requirit. Cum probatione methylationis multigenicae in hoc studio, ColonAiQ®, aegroti monitorium dynamicum ctDNA decima parte pretii habere et relationem intra duos dies accipere possunt.
Secundum 560,000 novos casus cancri colorectali in Sinis quotannis, aegroti clinici, praesertim cum cancro colorectali stadio II-III (proportio est circiter 70%), urgentiorem postulationem monitorii dynamici habent, cum magnitudo mercatus monitorii dynamici MRD cancri colorectali ad milliones hominum quotannis perveniat.
Patet eventus investigationis momentum scientificum et practicum magni momenti habere. Per studia clinica prospectiva magnae scalae, confirmatum est technologiam methylationis multigenicae ctDNA sanguinis, per PCR fundatam, ad praedictionem recidivationis cancri colorectali et monitorationem recidivationis adhiberi posse, cum sensibilitate, opportunitate et efficacia sumptuum, ita ut medicina accurata pluribus aegris cancro laborantibus melius prosit. Studium fundatur in ColonAiQ®, probatione methylationis multigenicae pro cancro colorectali a KUNY evoluta, cuius valor applicationis clinicae in examine et diagnosi praecoci per studium clinicum centrale confirmatus est.
Gastroenterologia (IF33.88), praecipua acta diurna internationalia in agro morborum gastrointestinalium anno 2021, eventus investigationum multicentricarum Nosocomii Zhongshan Universitatis Fudan, Nosocomii Cancri Universitatis Fudan, aliarumque institutionum medicarum auctoritativarum una cum KUNYAN Biological rettulit, quae egregiam ColonAiQ® ChangAiQ® efficaciam in examine et diagnosi praecoci cancri colorectali confirmaverunt, et initio applicationem potentialem in monitoratione prognosis cancri colorectali exploraverunt.
Ad ulterius comprobandum usum clinicum methylationis ctDNA in stratificatione periculi, directione de curationibus et monitoratione recidivationis praecocis in cancro colorectali stadii I-III, turma investigationis 299 aegros cum cancro colorectali stadii I-III inclusit, qui chirurgiam radicalem subierunt et exempla sanguinis in unoquoque puncto observationis (tribus mensibus inter se distantibus) intra unam hebdomadam ante chirurgiam, unum mensem post chirurgiam, et in therapia adjuvante postoperativa pro probatione dynamica ctDNA sanguinis collegerunt.
Primo, inventum est probationem ctDNA periculum recidivationis in aegris cancro colorectali mature praedicere posse, tam ante operationem quam post operationem mature. Aegri ctDNA praeoperativi probabilitatem recidivationis postoperativae maiorem habebant quam aegri ctDNA praeoperativi negativi (22.0% > 4.7%). Probatio ctDNA postoperativa praecox adhuc periculum recidivationis praedicebat: uno mense post resectionem radicalem, aegri ctDNA positivi 17.5 vicibus probabilius recidivationis erant quam aegri negativi; turma etiam invenit probationem ctDNA et CEA coniunctam leviter auxisse perfunctionem in detegenda recidivatione (AUC=0.849), sed differentia non erat significativa comparata cum sola probatione ctDNA (AUC=0.839). Differentia non erat significativa comparata cum sola probatione ctDNA (AUC=0.839).
Classificatio clinica cum factoribus periculi coniuncta nunc fundamentum principale est stratificationis periculi aegrotorum cancro laborantium, et in paradigma hodierno, numerus magnus aegrotorum adhuc recidivat [4], et urgente est necessitas meliorum instrumentorum stratificationis, cum curatio excessiva et insufficientis in clinica coexistant. Innixa hac re, turma aegrotos cum cancro colorectali stadio III in subgreges diversos classificavit, secundum aestimationem periculi recidivationis clinicae (periculum altum (T4/N2) et periculum humile (T1-3N1)) et periodum curationis adjuvantis (3/6 menses). Analysis invenit aegrotos in subgrege alto periculo aegrotorum ctDNA-positivorum inferiorem recidivationis ratem habuisse si sex menses therapiae adjuvantis accepissent; in subgrege humili periculo aegrotorum ctDNA-positivorum, nulla differentia significativa erat inter cyclum curationis adjuvantis et exitus aegrotorum; dum aegroti ctDNA-negativi significanter meliorem prognosin quam aegroti ctDNA-positivi et longiorem periodum postoperativam sine recidivatione (RFS) habuerunt; Cancer colorectali stadium I et stadium II periculi humilis. Omnes aegroti ctDNA-negativi nullam recidivationem intra biennium experti sunt; ergo, integratio ctDNA cum notis clinicis exspectatur ut stratificationem periculi ulterius optimizet et recidivationem melius praedicat.
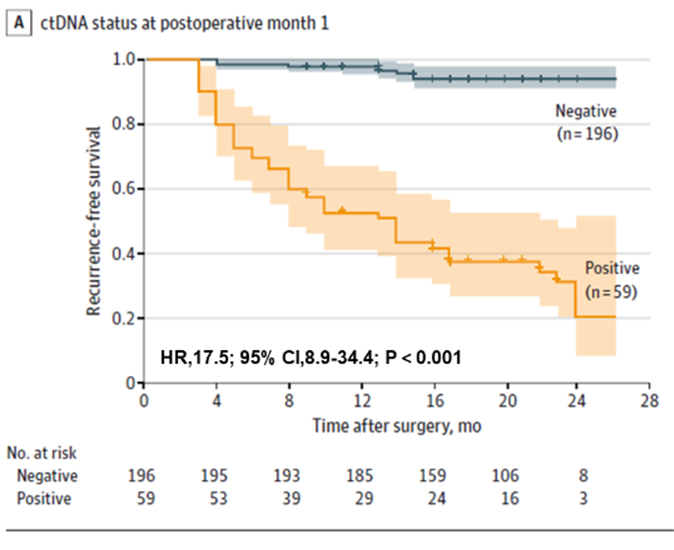
Figura 1. Analysis ctDNA plasmatica apud POM1 ad detectionem praecocem recidivationis cancri colorectalis.
Ulteriora experimenta dynamica ctDNA demonstraverunt periculum recidivationis significanter maius esse in aegris cum experimento ctDNA dynamico positivo quam in aegris cum ctDNA negativo per tempus monitorationis recidivationis morbi post curationem definitivam (post chirurgiam radicalem + therapiam adjuvantem) (Figura 3ACD), et ctDNA recidivationem tumoris usque ad 20 menses ante imagines indicare posse (Figura 3B), offerens possibilitatem detectionis praecocis recidivationis morbi et interventionis opportunae.
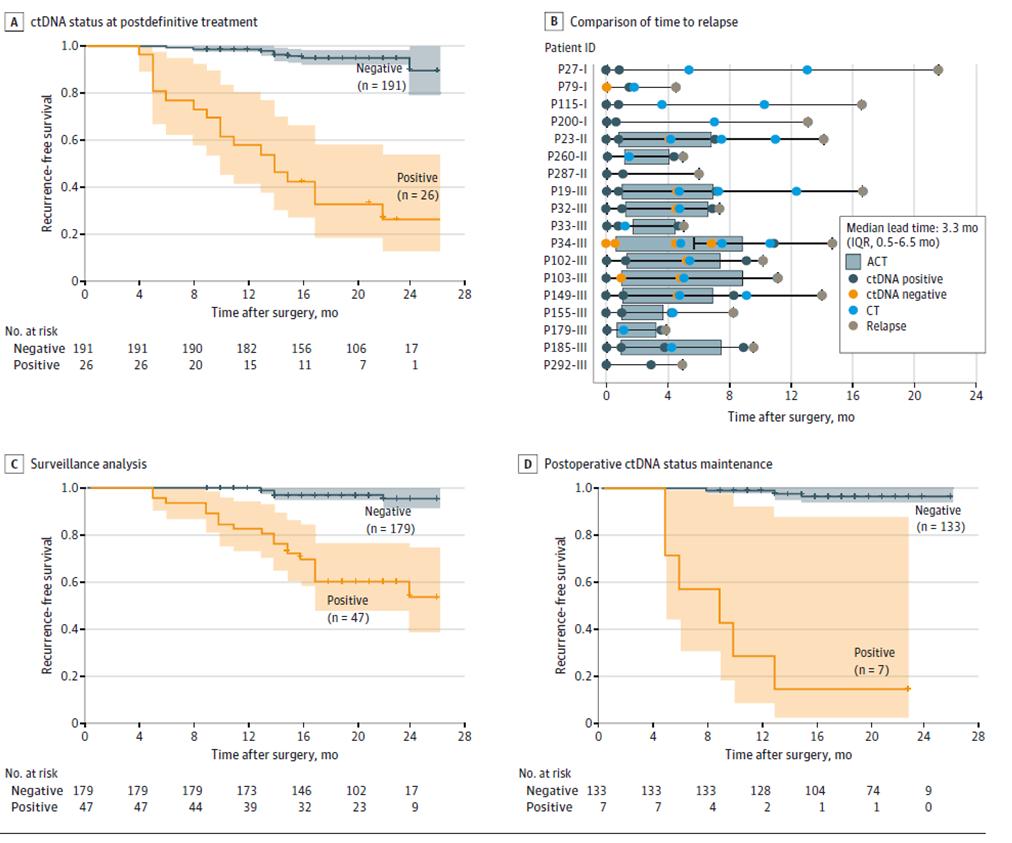
Figura 2. Analysis ctDNA secundum cohortem longitudinalem ad recidivationem cancri colorectalis detegendam.
"Magnus numerus studiorum medicinae translationalis in cancro colorectali disciplinam ducit, praesertim probatio MRD in ctDNA fundata magnum potentialem demonstrat ad curationem postoperativam aegrotorum cancro colorectali emendandam, per facultatem stratificationis periculi recidivationis, directionem consiliorum curationis et monitorationem recidivationis praecocis."
Commodum eligendi methylationem DNA ut novum indicem MRD prae detectione mutationis est quod non requirit examinationem sequentiationis totius genomi textuum tumoris, directe ad probationes sanguinis adhibetur, et vitat eventus falsos positivos propter detectionem mutationum somaticarum oriundarum ex textibus normalibus, morbis benignis, et haematopoiesi clonali.
Hoc studium aliaque studia conexa confirmant probationem MRD in ctDNA fundatam esse gravissimum factorem periculi independens pro recidivatione cancri colorectali stadii I-III et adhiberi posse ad adiuvandum decisiones de curatione dirigendas, inter quas "escalatio" et "degradatio" therapiae adjuvantis. MRD est gravissimus factor periculi independens pro recidivatione post chirurgiam pro cancro colorectali stadii I-III.
Campus MRD celeriter evolvitur cum pluribus probationibus novis, valde sensibilibus et specificis, in epigenetica (methylatione DNA et fragmentomica) et genomica (sequentiatione ultra-profunda directa vel sequentiatione totius genomi) fundatis. Exspectamus ColonAiQ® studia clinica magnae scalae ordinare pergere et novum indicem probationis MRD fieri posse, quod accessibilitatem, altam efficaciam et pretium moderatum coniungit et late in praxi clinica cotidiana adhiberi potest.
Referentiae
[1] Mo S, Ye L, Wang D, Han L, Zhou S, Wang H, Dai W, Wang Y, Luo W, Wang R, Xu Y, Cai S, Liu R, Wang Z, Cai G. Detectio Praecox Morbi Residui Molecularis et Stratificatio Periculi pro Cancro Colorectali Stadii I ad III per Methylationem DNA Tumoris Circulantis. JAMA Oncol. 2023 Apr 20.
[2] "Onus morbi cancri colorectalis in populatione Sinensi: num mutatum est annis proximis?", *Acta Epidemiologica Sinensia*, Vol. 41, No. 10, Octobris 2020.
[3] Tarazona N, Gimeno-Valiente F, Gambardella V, et al. Sequentiatio DNA tumoris circulantis novae generationis ad morbum residuum minimum in cancro coli localizato investigandum. Ann Oncol. 1 Nov. 2019;30(11):1804-1812.
[4] Taieb J, André T, Auclin E. Expolitio therapiae adiuvantis pro cancro coli non metastatico, novae normae et prospectus. Cancer Treat Rev. 2019;75:1-11.
Tempus publicationis: XXVIII Aprilis MMXXIII
 中文网站
中文网站