Primo die XXIX Decembris, NEJM novum studium clinicum phasis III novi coronaviri Sinensis VV116 in interrete divulgavit. Resultata demonstraverunt VV116 non peiorem fuisse quam Paxlovid (nematovirum/ritonavirum) quod ad durationem convalescentiae clinicae attinet et pauciores eventus adversos habuisse.
Fons imaginis: NEJM
Medianum tempus convalescentiae 4 dies, proportio eventuum adversorum 67.4%
VV116 est medicamentum nucleosidicum orale contra novum coronavirus (SARS-CoV-2) elaboratum una cum Junsit et Wang Shan Wang Shui, et est inhibitor RdRp una cum remdesivir Gilead, molnupiraviro Merck Sharp & Dohme et azelvudino Real Biologics.
Anno MMXXI, experimentum clinicum secundae phasis VV116 in Uzbecia completa est. Resultata studii demonstraverunt gregem VV116 symptomata clinica melius emendare et periculum progressionis ad formam criticam et mortis significanter reducere potuisse, comparatione cum grege testigo. Ob positivos eventus huius experimenti, VV116 in Uzbecia ad curationem aegrotorum cum COVID-19 moderato ad gravem approbatum est, et primum novum medicamentum coronarium orale ad venditionem transmarinam in Sinis approbatum factum est [1].
Hoc experimentum clinicum phasis III[2] (NCT05341609), a Professore Zhao Ren Hospitalis Shanghaiensis Ruijin, Professore Gaoyuan Hospitalis Shanghaiensis Renji et Academico Ning Guang Hospitalis Shanghaiensis Ruijin ductum, peractum est durante eruptione a variante Omicron (B.1.1.529) a Martio ad Maium Shanghaii causata, eo consilio ut efficaciam et salutem VV116 contra Paxlovid ad curationem praecocem aegrotorum cum COVID-19 levi ad moderatum aestimaret. Propositum erat efficaciam et salutem VV116 contra Paxlovid ad curationem praecocem aegrotorum cum COVID-19 levi ad moderatum aestimare.
Fons imaginis: Referentia 2
Experimentum clinicum multicentricum, observatore caeco, aleatorium, et moderatum, 822 aegrotorum adultorum Covid-19, qui periculo progressionis alto et symptomatibus levibus vel moderatis obnoxii erant, inter diem quartum Aprilis et diem secundum Maii anni 2022 peractum est ad idoneitatem participantium ex septem nosocomiis Shanghaii, Sinarum, aestimandam. Tandem, 771 participes vel VV116 (384, 600 mg singulis 12 horis die 1 et 300 mg singulis 12 horis diebus 2-5) vel Paxovid (387, 300 mg nimatuvir + 100 mg ritonaviri singulis 12 horis per 5 dies) per os acceperunt.
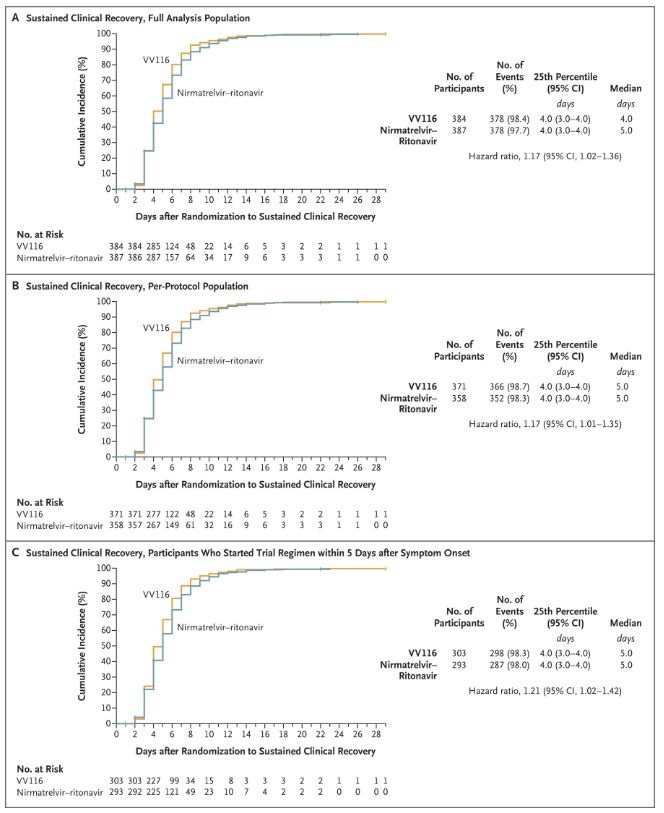
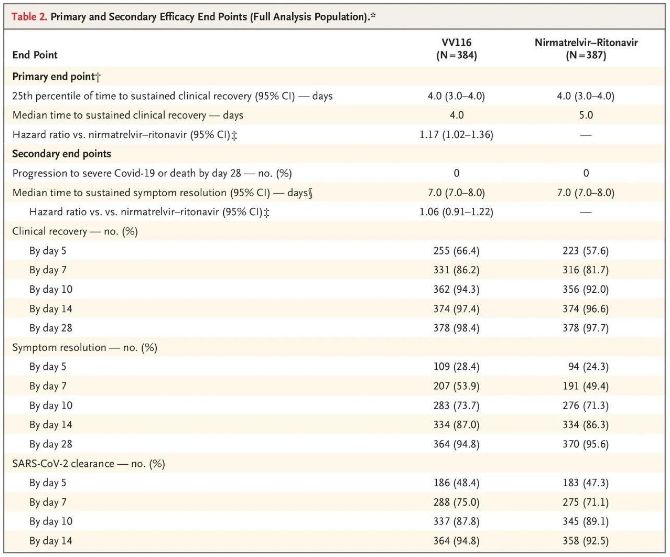
Eventus huius studii clinici demonstraverunt curationem praecocem cum VV116 pro COVID-19 levi vel moderato finem primarium (tempus ad recuperationem clinicam sustentam) a protocollo clinico praedictum attigisse: tempus medianum ad recuperationem clinicam erat 4 dies in grege VV116 et 5 dies in grege Paxlovid (proportio periculi, 1.17; 95% CI, 1.02 ad 1.36; limes inferior. >0.8).
Tempus recuperationis clinicae conservandum
Finis efficaciae primarii et secundarii (analysis comprehensiva populationis)
Fons imaginis: Referentia 2
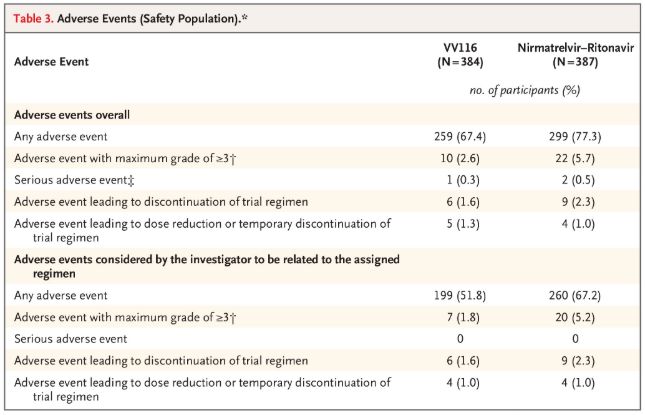
Quod ad salutem attinet, participes qui VV116 acceperunt pauciores eventus adversos (67.4%) quam qui Paxlovid acceperunt (77.3%) post 28 dies nuntiaverunt, et incidentia eventuum adversorum Gradus 3/4 minor erat pro VV116 (2.6%) quam pro Paxlovid (5.7%).
Eventus adversi (homines tuti)
Fons imaginis: Referentia 2
Controversiae et quaestiones
Die XXIII mensis Maii, anno MMXXII, Juniper patefecit studium clinicum registrationis Phase III VV116 contra PAXLOVID ad curationem praecocem COVID-19 levis vel moderati (NCT05341609) finem primarium studii attigisse.
Fons imaginis: Referentia 1
Tempore quo singula de experimento desunt, controversia circa studium Phase III duplex erat: primo, studium simplex caecum erat, et, absente comparatione placebo, timebatur ne difficile esset medicamentum omnino obiective iudicare; secundo, quaestiones de finibus clinicis ortae sunt.
Criteria inclusionis clinica pro *Juniper* sunt (i) eventus positivi pro novo examine coronae, (ii) unum vel plura symptomata COVID-19 levia vel moderata, et (iii) aegroti periculo alto COVID-19 gravis, incluso morte, obnoxii. Tamen, solum finis clinicus primarius est "tempus ad recuperationem clinicam sustentam".
Paulo ante nuntium, die XIV mensis Maii, Juniper fines clinicos reviderat, unum ex primariis finibus clinicis, "proportionem conversionum ad morbum gravem aut mortem" [3], removendo.
Fons imaginis: Referentia 1
Hae duae principales contentionis puncta etiam in studio edito specifice tractatae sunt.
Ob subitam eruptionem Omicronis, productio tabularum placebo pro Paxlovid non completa erat ante initium experimenti, et ideo investigatores hoc experimentum consilio dupliciter caeco et dupliciter simulato peragere non potuerunt. Quod ad aspectum simplex caecum experimenti clinici attinet, Juniper dixit protocollum post communicationem cum auctoritatibus moderatoribus peractum esse et consilium simplex caecum significare neque investigatorem (incluso aestimatore finis studii) neque sponsorem specificam allocationem medicamenti therapeutici cognituros esse donec finalis database clausa sit ad finem studii.
Usque ad tempus analysis finalis, nullus ex participibus in experimento mortem aut progressionem ad gravem morbum Covid-19 expertus erat, itaque nullae conclusiones de efficacia VV116 in prohibenda progressione ad gravem vel criticum Covid-19 aut mortem duci possunt. Data indicaverunt tempus medianum aestimatum a randomizatione ad regressionem sustentatam symptomatum Covid-19-relatorum fuisse 7 dies (95% CI, 7 ad 8) in utroque grege (proportio periculi, 1.06; 95% CI, 0.91 ad 1.22) [2]. Non difficile est explicare cur finis primarius 'ratio conversionis ad gravem morbum aut mortem', qui initio ante finem experimenti constitutus erat, sublatus sit.
Die XVIII mensis Maii anni MMXXII, acta diurna "Emerging Microbes & Infections" eventus primi experimenti clinici VV116 in aegris infectis variante Omicron [4] divulgaverunt, studii cohortis aperti, prospectivi cum CXXXVI aegris in nosocomio confirmatis.
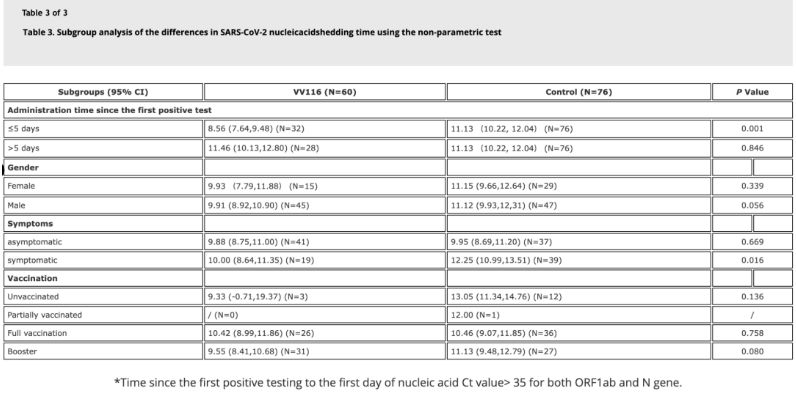
Data ex studio demonstraverunt aegros infectione Omicron affectos, qui VV116 intra quinque dies a primo examine positivo acidi nucleici usi sunt, tempus ad regressionem acidi nucleici 8.56 dierum habuisse, minus quam 11.13 dies in grege comparationis. Administratio VV116 aegris symptomaticis intra tempus huius studii (duobus ad decem diebus a primo examine positivo acidi nucleici) tempus ad regressionem acidi nucleici in omnibus aegris minuit. Quod ad salutem medicamenti attinet, nulli effectus adversi graves in grege curationis VV116 observati sunt.
Fons imaginis: Referentia 4
Tria experimenta clinica de VV116 in progressu sunt, quorum duo sunt studia phasis III de COVID-19 levi ad moderatum (NCT05242042, NCT05582629). Alterum experimentum pro COVID-19 moderato ad gravem est studium clinicum phasis III internationale, multicentricum, aleatorium, dupliciter caecum (NCT05279235) ad efficaciam et salutem VV116 comparatam cum curatione ordinaria aestimandam. Secundum nuntium a Juniper factum, primus aegrotus conscriptus et dosis data est mense Martio 2022.
Fons imaginis: clinicaltrials.gov
Referentiae:
[1] Junshi Biotech: Nuntiatio de fine principali studii clinici Phase III registrati VV116 contra PAXLOVID ad curationem praecocem COVID-19 levis vel moderati.
[2] https://www.nejm.org/doi/full/10.1056/NEJMoa2208822?query=featured_home[3] https://clinicaltrials.gov/ct2/show/record/NCT05341609 [4] Ensi Ma, Jingwen Ai, Yi Zhang, Jianming Zheng, Xiaogang Gao, Junming, Xu, Liogang Gao, Junming, Xu Liogang Gao Sol iacentes, Heyu Huang, Quanbao Zhang, Linlin Xu, Yanting Jin, Rui Chen, Guoyue Lv, Zhijun Zhu, Wenhong Zhang, Zhengxin Wang. (2022) Infectiones Omicron profile et vaccinationis status inter iecoris 1881 recipientes transferunt: cohors multi-centra retrospectiva. Microbes & Infectiones 11:1, pages 2636-2644.
Tempus publicationis: VI Ianuarii MMXXIII
 中中 网站
中中 网站